Product Technology Overview: Cutimed® Off-Loader Select Total Contact Cast System
December 18, 2014
According to Centers for Disease Control and Prevention (CDC), the number of adults between the ages of 18-79 with newly diagnosed diabetes has more than tripled in the last 30 years. Foot ulcers are a major complication of uncontrolled diabetes, and 25% of the patients will be affected with foot ulcers in their lifetime, the majority of which lead to lower extremity amputations.
The basis of treatment for diabetic foot ulcers is dependent on three major factors: vascular status, infection control, and offloading the pressure site. Major contributing factors in development of ulcers are increased localized pressure and sensory loss. Hence, these two factors are the focus of treatment for successful healing of an ulcer. Total Contact Cast (TCC) systems are the gold standard for offloading a neuropathic ulcer however, according to Fife et al, only 6% of clinicians utilize it for offloading on a regular basis, with a fear of complications and lack of training in its application as the main reasons why they did not.
Total Contact Casting for Effective Offloading
The effectiveness of the TCC is dependent on pressure distribution and load transfer. The anatomically molded cast distributes force evenly across the entire plantar surface of the foot thereby reducing plantar pressures concentrated in the area of the ulceration. At the same time, vertical forces on the foot are transferred to the lateral walls of the conical leg section of the cast. Research has shown that approximately 30-40% of the force directed on the foot is absorbed by the walls of the cast.
In addition to alleviating pressure, the TCC forces the patient to stay compliant with treatment. TCC and non-removable cast walkers have been demonstrated to be the best modalities for offloading the foot. The second you allow the patient to remove the device for sleeping, bathing, and dressing changes you reduce the general effectiveness from 80-90% healing in 12 weeks to 50-60%. TCCs reduce patient mobility thus reducing the impact from walking on the open ulcer. Wound healing is significantly delayed if there is repetitive trauma from ambulation. Moreover, casting contributes to edema reduction, and protects the ulcer from external contamination.
There are several Total Contact Cast kits that are commercially available today to make it easier for clinicians to utilize this therapy. Kits reduce the preparation and application time and also reduce the cost to the clinic in both aggregating the materials and the time for application. Kits range from very traditional components with minimal padding and a rubber rocker walker for ambulation to padded roll-on fiberglass models with fairly sophisticated removable walkers to use when walking.
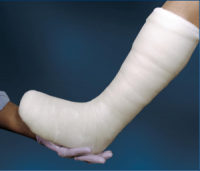 The Cutimed® Off-Loader Select is neither and is used in our clinic for several reasons. Only a light or minimally padded rigid cast has the ability to truly transfer weight from the foot to the leg section of the cast. We have found other kits on the market to be too flexible in many cases to manage patients over 250 pounds without extra cast tape for reinforcement. Extra rolls of cast tape add to the cost of the device and making it less economical.
The Cutimed® Off-Loader Select is neither and is used in our clinic for several reasons. Only a light or minimally padded rigid cast has the ability to truly transfer weight from the foot to the leg section of the cast. We have found other kits on the market to be too flexible in many cases to manage patients over 250 pounds without extra cast tape for reinforcement. Extra rolls of cast tape add to the cost of the device and making it less economical.
In addition to the Cutimed® Off-Loader Select's rigidity, it also uses a low profile cast shoe for its walking base. Having a removable cast shoe was important to us because many patients complained about having to get into bed with a dirty rubber rocker. They also complained of back pain and gait instability associated with the limb length discrepancy caused by the rubber rocker.
Application of the Total Contact Cast System
An experienced clinician or cast technician can apply a Cutimed® Off-Loader Select in 10 minutes. All the necessary materials are included in the kit. You begin by preparing the patient with your selected wound dressing (a foam dressing is provided in the kit). The limb is covered with their own stockinette and precut felt padding included in the kit is applied. The toes are covered with a fenestrated adhesive foam and light cast padding applied as per the clinicians preference. The fiberglass casting material is then rolled on with the foot held at 90 degrees to the ankle with a casting board provided with the first order of kits. A rocker bottom cast shoe is then applied and the patient gait trained to be sure they do not need additional ambulatory aides to walk. Our clinicians independently self-selected this cast kit over several others available during the testing period.
To learn more about this company and product visit http://www.woundsource.com/company/bsn-medical-inc
Sources:
1. Fife CE, Carter MJ, Walker D. Why is it so hard to do the right thing in wound care? Wound Repair and Regeneration. 2010;18(2):154-8. doi: 10.1111/j.1524-475X.2010.00571.x.
2. Greenhagen RM, Wukich DK. Total Contact Casting for Neuropathic Ulcers: A Lost Art? The Journal of Diabetic Foot Complications 2009;1(4):85-93. http://jdfc.org/wp-content/uploads/2010/09/Volume1Issue4-Total_contact_…
3. Mueller MJ, Diamond JE, Sinacore DR, et al. Total contact casting in treatment of diabetic plantar ulcers. Controlled clinical trial. Diabetes Care. 1989;12(6):384-8.
About the Authors:
Rizwan Tai is a third year podiatric medical student at Temple University in Philadelphia, Pennsylvania. He graduated from the University of Houston in 2011 with a Bachelor of Business Administration and a minor in Biology. His interest in podiatry was sparked after a close relative of his suffered from amputations of toes secondary to diabetic complications, and his mother suffering from plantar fascia pain.
Dr. James McGuire is the director of the Leonard S. Abrams Center for Advanced Wound Healing and an associate professor of the Department of Podiatric Medicine and Orthopedics at the Temple University School of Podiatric Medicine in Philadelphia.
The views and opinions expressed in this blog are solely those of the author, and do not represent the views of WoundSource, HMP Global, its affiliates, or subsidiary companies.
Have a product to submit?
Be included in the most comprehensive wound care products directory
and online database.
Learn More









Follow WoundSource
Tweets by WoundSource