Amniotic Membrane Explained: What it is and How it is Used in Wound Care
August 19, 2015
The human amniotic membrane has been used in wound care for almost a century. It is now enjoying a resurgence in use in wound care, thanks to new techniques that allow this unique material to be dried and stored for prolonged periods of time.
Structure and Function of the Amniotic Membrane
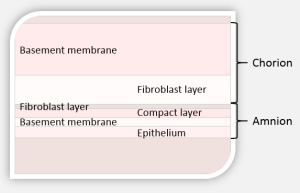
The amniotic membrane surrounds and protects the developing fetus in utero and separates mother and fetus. If you were to look closely at the amniotic membrane, you would be able to notice that the membrane is comprised of several layers. The membrane can be easily separated into two distinct layers: the amnion layer and the chorion layer, which are separated by a jelly-like layer. The amnion layer of the membrane, or fetal side, has a layer of epithelial cells which can easily be removed with simple cell scraping, revealing a see-through underlying layer. The chorion layer is the maternal side of the amniotic membrane. Both layers have a basement membrane and a stromal layer (Medscape, 2012).
If you looked at the amniotic membrane under a microscope, you would be able to appreciate three different types of material: collagen and extracellular matrix, biologically active cells and regenerative molecules. The extracellular matrix provides structure and contains a number of specialized proteins, including proteoglycans, fibronectin, laminins and others. Several types of collagen add structural strength to the membrane. The biologically active cells include stem cells, which function to regenerate new cellular materials within the lining of the membrane. Fibroblasts help to strengthen the tissue, and epithelial cells aid in the healing process via receptors on the cell surface. Regenerative molecules, which are important for growth and healing, are present in the amniotic membrane as well. These include numerous types of growth factors such as fibroblast growth factors, platelet-derived growth factors, metalloproteinases and others. Immunosuppressive cytokines prevent the amniotic membrane from being seen as 'foreign' by both the mother and infant's immune systems. There are also a number of other specialized molecules, such as defensins which protect against bacterial infection (Medscape, 2012).
Wound Healing Properties of Amniotic Membrane
Amniotic membrane has a number of characteristics that make it especially suited to wound healing. The amniotic membrane:
- contains a significant number of cytokines and essential growth factors
- reduces pain when applied to a wound
- increases and enhances the wound healing process
- has antibacterial properties
- is non-immunogenic (will not be seen as foreign material)
- provides a biological barrier
- provides a matrix for migration and proliferation of cells
- reduces inflammation
- reduces scar tissue formation
Dehydration of Amniotic Membrane Tissue
In the past, amniotic tissue was sterilized and stored at 4°C. Amniotic tissue could only be used for up six weeks, at which point it was no longer useful. Now, this material can be cleansed, dehydrated and sterilized, which means that the shelf life of amniotic membrane has been greatly increased.
Indications for Use
What types of wounds can amniotic membrane be used on? Traditionally, amniotic membrane has been used on burns; nowadays, however, amniotic membrane can be used on a wide variety of wounds. It is important to note that amniotic membrane should be used only after conservative treatment has failed. In other words, amniotic membrane may be used for wounds that are chronic and non-healing.
Application of Amniotic Membrane
Prior to using amniotic membrane, a thorough initial assessment of the wound is necessary, as is a medical history. Gather information on the history of the wound, including duration, what treatments have been tried and patient comorbidity. Document wound appearance, size, depth, presence of necrotic tissue and note whether bone or other structures are visible. Assess circulatory status, nutrition and other barriers to healing. These are the same steps that you would follow prior to implementing any wound treatment and are not specific to amniotic membrane treatment.
Next, prepare the wound bed by performing any necessary debridement. The wound bed should be clear of any necrotic tissue and should not have any signs of infection. Amniotic membrane is supplied in a sterile container, and sterile scissors may be used to cut a piece of the amniotic membrane to fit the wound. The material may be applied wet or dry. Note that the stromal collagen layer must be facing the wound - read the manufacturer's recommendations to determine how to apply the material. You can use steri-strips to hold the graft in place. There is no need to suture the material in place. A secondary dressing which promotes moist wound healing should be chosen as a secondary dressing. The graft should not be disturbed for at least one to two weeks.
After one to two weeks the amniotic membrane allograft will be incorporated into the wound. You should begin to see improvement in the wound in terms of size and depth within 2 to 3 weeks, or even sooner. You can apply a second graft once slowing of wound healing has occurred, as typically observed by wound measurements over time (Podiatry Today, 2015).
The use of amniotic membrane in the management of chronic wounds is an exciting new development which provides another option for wounds that fail to heal using traditional wound therapies and dressings.
Sources:
Fetterolf D, Snyder R. Scientific and Clinical Support for the Use of Dehydrated Amniotic Membrane in Wound Management. Wounds. 2012;24(10):299-307.
Zelen C, Serena T. Amniotic Membrane: Can it facilitate healing? Podiatry Today. 2015 Apr;28(4). Available at http://www.podiatrytoday.com/amniotic-membrane-can-it-facilitate-healing
About The Author
Laurie Swezey RN, BSN, CWOCN, CWS, FACCWS is a Certified Wound Therapist and enterostomal therapist, founder and president of WoundEducators.com, and advocate of incorporating digital and computer technology into the field of wound care.
The views and opinions expressed in this content are solely those of the contributor, and do not represent the views of WoundSource, HMP Global, its affiliates, or subsidiary companies.