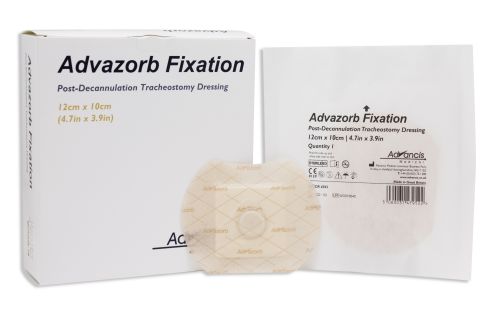
Advazorb Fixation® Tracheostomy Dressing
Advazorb Fixation® Tracheostomy Dressing is a hydrophilic foam dressing with a soft silicone contact layer and surrounding adhesive.
Advancis Medical
Advancis Medical is a UK-based medical device company building an international reputation for developing, manufacturing, and marketing a range of advanced wound care products. medi® USA sells and distributes Advancis products in the United States.
Benefits
• Supplied sterile and individually wrapped, ready for use
• Location button enables accurate identification of tracheostomy site
• Maintains a moist environment and manages exudate
• Allows atraumatic dressing change
• Provides a barrier from bacteria
Indications
Advazorb Fixation® Tracheostomy Dressing is for use on stoma sites after decannulation.
Contraindications
Contraindicated for use on arterial bleeds and heavily bleeding wounds. Do not use if allergic to silicone, foam, or polyurethane.
Storage Requirements
Store at room temperature, out of direct sunlight and keep dry.
How Supplied/Sizing
Product features
Recommended Use
Stoma Sites After Decannulation
Mode of Use/Application
Clean and dry stoma site. Peel center backing off dressing. Holding the PVC button, place the dressing directly over the stoma site. Peel off remaining backing strips, and press firmly over skin.
Removal & Change Frequency
Change dressing every 5 to 7 days or when pad swells due to absorption.
Clinically Tested
CHG-compatible
Does not contain natural rubber latex
Latex-friendly
Non-allergenic
Non-comedogenic
Non-cytotoxic
Non-irritating
No posters match your selected filters. Remove some filtres, or reset them and start again.
Have a product to submit?
Be included in the most comprehensive wound care products directory
and online database.
Learn More










