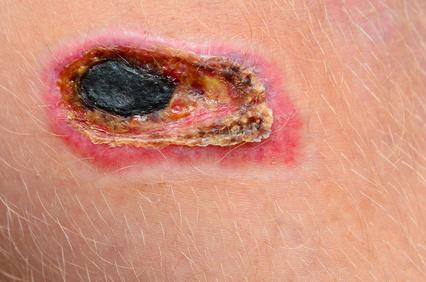
Biofilm and Wound Healing
January 1, 2018
Composition of Biofilm
Biofilm is a complex microbial community containing self- and surface-attached microorganisms that are embedded in an extracellular polymeric substance.1,2 The extracellular polymeric substance is a primarily polysaccharide protective matrix synthesized and secreted by the microorganisms that attaches the biofilm firmly to a living or non-living surface. This protective covering does not allow the body's immune system to recognize the presence of the microorganism; therefore, the bacteria evade an immune response, avoid detection by standard diagnostic techniques, and avoid destruction by standard treatments.3 Because of the tenacity of the attached biofilm, the microoganisms are able to resist physical forces, such shear, and are able to withstand nutrient and moisture deprivation, altered pH, and the impact of antibiotics and antiseptics. For the purposes of this discussion we break down the formation and actions of biofilms and discuss their impact on wound healing.
Relationship of Biofilm Formation with Delayed Wound Healing
Before 2008, when bacterial overload interfered with wound healing it was typically referred to as critical colonization. A hypothesis released in 2008 discussed the relationship of biofilm with delayed wound healing, and this hypothesis spurred a plethora of research on biofilm in the last decade.4 These studies examined the correlation of recalcitrant wounds and the existence of biofilm and revealed that most of these wounds contained biofilm.
Ubiquity and Formation of Biofilm
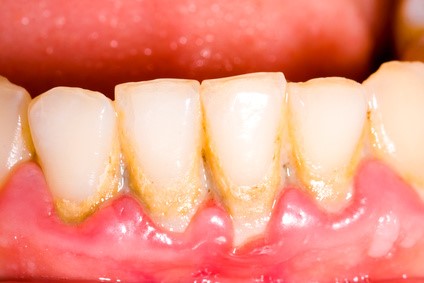
Biofilms are found not only in wounds, but also on medical devices such as prosthetic heart valves, orthopedic implants, intravascular catheters, and artificial hearts, to name a few. More familiar examples of biofilms include those adhered to rocks at the bottom of a stream, on the floors of showers, and in sewage pipes, sometimes causing blockage and corrosion. Teeth with dental plaque also have biofilm, which can lend to the development of tooth decay and gum disease.2
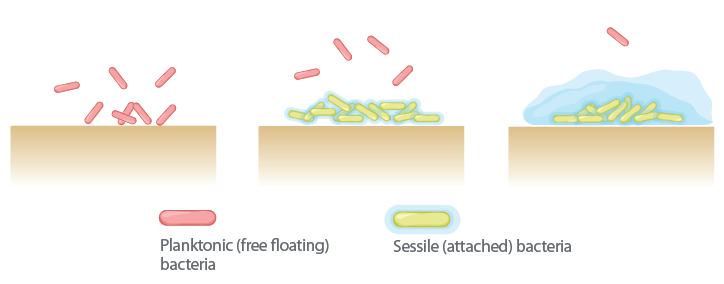
Biofilm is present in 60%-90% of chronic wounds and in only 6% of acute wounds. Biofilm formation contributes to chronic inflammation, delayed healing, and infection.5-7 So how does this happen? Biofilm forms in three stages. Despite the common thought that microorganisms are all free floating, most tend to attach to surfaces and form biofilms, and in stage 1 this attachment is reversible. The attachment can form in minutes, create microcolonies within four hours, and become biocide tolerant within 12 hours. With stage 2 the microorganisms have become firmly attached to the wound bed, and the biofilm community begins to communicate triggering genetic changes to increase the chance of survival. In stage 3 the protective polysaccharide coating is fully formed. Within two to three days, depending on the species and environmental conditions, if disrupted these colonies can rapidly recover from disruption.
The process from biofilm recovery to being entirely reformed can take only 24 hours.6 The slimy coating of biofilm, in addition to polysaccharides, also contains proteins, glycolipids, and both living and dead bacterial DNA. A chronic inflammatory response is initiated, which increases exudates, neutrophils, and macrophages, host cell secretion of matrix metalloproteases or MMPs, and reactive oxygen species. These activities are stimulated at the cellular level by the host to break down the attachment the biofilm has developed that secures it to the wound bed, but despite their efforts, these activities are not completely effective in achieving this feat because of the tenacity of the biofilm. In simple terms, biofilm can be described as microorganisms living in a thick, slimy barrier of sugars and proteins that protect them from external threats.6
The prolonging of this process and the continued secretion of these substances, in turn, can damage normal and healing tissue.2 These fully mature biofilms keep shedding planktonic bacteria microcolonies and fragments of biofilm, which attach to other parts of the wound bed and facilitate the spread of biofilm.4
When you think of a community, you think of its members utilizing their collective strengths to support the community as a whole. Mixed microbial communities do just that and utilize their strengths and abilities to further the survival of the group, an approach that gives significant protective advantages. Conditions that predispose wounds to developing biofilm are not completely understood. That said, conditions that impede the effectiveness of antibiotics or suppress the immune system may support the development of biofilm. Examples of such conditions include ischemia, poor nutrition, necrosis, and comorbid conditions.
Public Health Impact of Biofilm in Wound Healing
A substantial body of evidence that has emerged over the last decade shows that at least half of all chronic non-healing wounds contain biofilm, which has a role in the delay of healing. This issue could be contributing to elevated health care costs to the tune of billions of dollars globally.4 Understanding the development of biofilm will aid clinicians in accurately assessing wounds and developing strategies to identify its existence earlier to keep wound healing on a positive trajectory, thus saving clinical and financial resources and improving outcomes.
References:
1. Barker JC, Khansa I, Gordillo G. A formidable foe is sabotaging your results: What you should know about biofilms and wound healing. Plast Reconstr Surg. 2017;139(5):1184-94.
2. Hurlow J. Understanding biofilm: what a community nurse should know. Br J Community Nurs. 2016;21(9):26-33.
3. Song T, Duperthuy M, Wai SN. Sub-optimal treatment of bacterial biofilms. Antibiotics. 2016;5(2):23.
4. Metcalf DG, Bowler PG. Biofilm delays wound healing: a review of the evidence. Burns Trauma. 2013;1(1):5-12.
5. Percival SL, McCarty SM, Lipsky B. Biofilms and wounds: an overview of the evidence. Adv Wound Care (New Rochelle). 2015;4(7):373-81.
6. Phillips PL, Wolcott RD, Fletcher J, Schultz G.S. Biofilms made easy. Wounds Int. 2010;1(3). Available at: http://www.woundsinternational.com/made-easys/view/biofilms-made-easy
7. Stechmiller JK, Schultz G. Implementing Biofilm and Infection 2014 Guidelines. National Pressure Ulcer Advisory Panel.
The views and opinions expressed in this content are solely those of the contributor, and do not represent the views of WoundSource, HMP Global, its affiliates, or subsidiary companies.