Organogenesis
Organogenesis is a global leader in advanced wound care, offering a comprehensive portfolio of regenerative medicine products capable of supporting patients from early in the wound healing process through to wound closure, regardless of wound type.
Benefits
• Novel LayerLoc™ preservation method retains all layers of the placental membrane that contain numerous growth factors and cytokines
• Real-world evidence demonstrated that Nushield®, used as a protective wound covering, helps support an optimal environment for healing in a variety of wound types and sizes
Indications
NuShield® is an allograft intended for use in the management of acute and chronic wounds as well as appropriate surgical applications. NuShield® may be applied as a wound covering to a variety of partial- and full-thickness acute and chronic wounds and wounds with exposed tendon, muscle, joint capsule and bone. NuShield® can be applied from the onset and for the duration of the wound, weekly or at the discretion of the health care practitioner.
Contraindications
NuShield® is contraindicated for use on clinically infected wounds.
NuShield® is contraindicated for surgical implantation sites with active or latent infection.
Please refer to the NuShield® Instructions for Use for more information.
Warnings and Precautions
As with all allogeneic materials, it is not possible to provide an absolute guarantee that no infectious disease will be transmitted. However, this risk is greatly reduced by using processing treatments shown to be capable of reducing this risk as well as the use of strict donor screening criteria, laboratory testing, aseptic processing and terminal gamma irradiation of final product. Single patient, single use only. Do not sterilize or re-sterilize.
Adverse Effects/Reactions
Transmission of diseases of unknown etiology; transmission of known infectious agents including, but not limited to, viruses, bacteria and fungi; immune rejection of implanted HCT/P; or loss of function and/or integrity of implanted HCT/P due to resorption, fragmentation and/or disintegration.
Storage Requirements
Store at ambient temperature.
How Supplied/Sizing
HCPCS Code
Product features
Other features
Mode of Use/Application
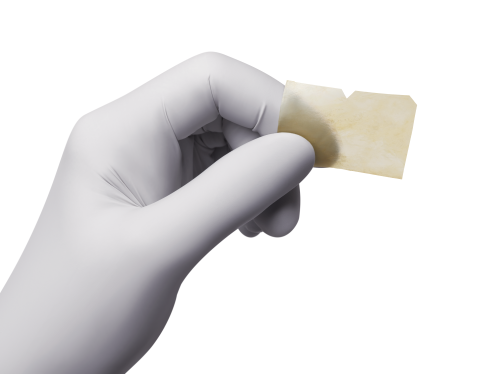
Remove NuShield® from its sterile packaging using aseptic technique, being careful to maintain the sterility of the product. To determine which side of NuShield® is the chorion side (to be placed in contact with the wound bed), verify that the middle notch is at the top, and the notched corner is to its right, then the upper side is epithelial and the side facing down is chorion. Place NuShield® onto the wound bed, being careful to preserve orientation of the product and ensure that the chorion side is in contact with the wound bed. Once NuShield® is placed, the product may be rehydrated as necessary, based on the characteristics of the wound (NuShield® is easier to handle prior to rehydration).
Additional Recommended Dressings
A non-adherent, non-occlusive primary dressing may be used directly over NuShield®.
No posters match your selected filters. Remove some filtres, or reset them and start again.
Have a product to submit?
Be included in the most comprehensive wound care products directory
and online database.
Learn More