Supporting Wound Closure With Cellular and/or Tissue-Based Products
February 28, 2022
Introduction
Cellular and/or tissue-based products (CTPs) comprise an exciting and emerging technology in wound care. Deep and large wounds require more than just a standard dressing, topical ointments, and time. Wounds that penetrate the reticular dermis and below (deeper than ~0.57 mm) require additional scaffolding to support wound closure and prevent significant scarring.1 CTPs provide this scaffolding as well as naturally occurring chemicals and cells that support wound healing.
Cellular and/or Tissue-Based Products in Wound Management
Currently, research supports the use of CTPs in patients with diabetic foot ulcers, venous leg ulcers, and burns.1 CTP use has been found to be a cost-effective method of promoting wound closure and healing when these products are used on wounds of appropriate severity.2 Wounds that do not respond to other methods of healing may benefit from the use of CTPs, despite the high initial cost of these treatments. CTPs help heal the wound before costly complications arise and aid in wound healing before long-term treatment is required.
The literature suggests the following essential properties of CTPs1:
- Absence of toxicity
- Absence of antigens
- Cost-effectiveness
- Ease of application
- Long shelf life
- Provision of a barrier against infection
- Cause of no more than minimal inflammation
- Improved wound healing
- Resistance to mechanical forces
However, no single CTP can currently meet all these suggested requirements. A combination of treatments will likely be required.
Living Versus Nonliving Products
CTPs are available in the form of living, nonliving, or biosynthetic tissues. In 2016, the American Standards for Testing and Materials (ASTM) International group3 published a classification for CTPs that includes biosynthetic, nonliving, and living as major categories. The main differences among these types of CTPs are their origin of production and the level of processing. Current literature does not include evidence on which types may be most effective for wound healing. However, different products currently have different levels of approval in the United Stated under the Food and Drug Administration1 and in other countries under their regulatory bodies.4
Cellular and/or Tissue-Based Products as Scaffolding to Aid in Wound Healing
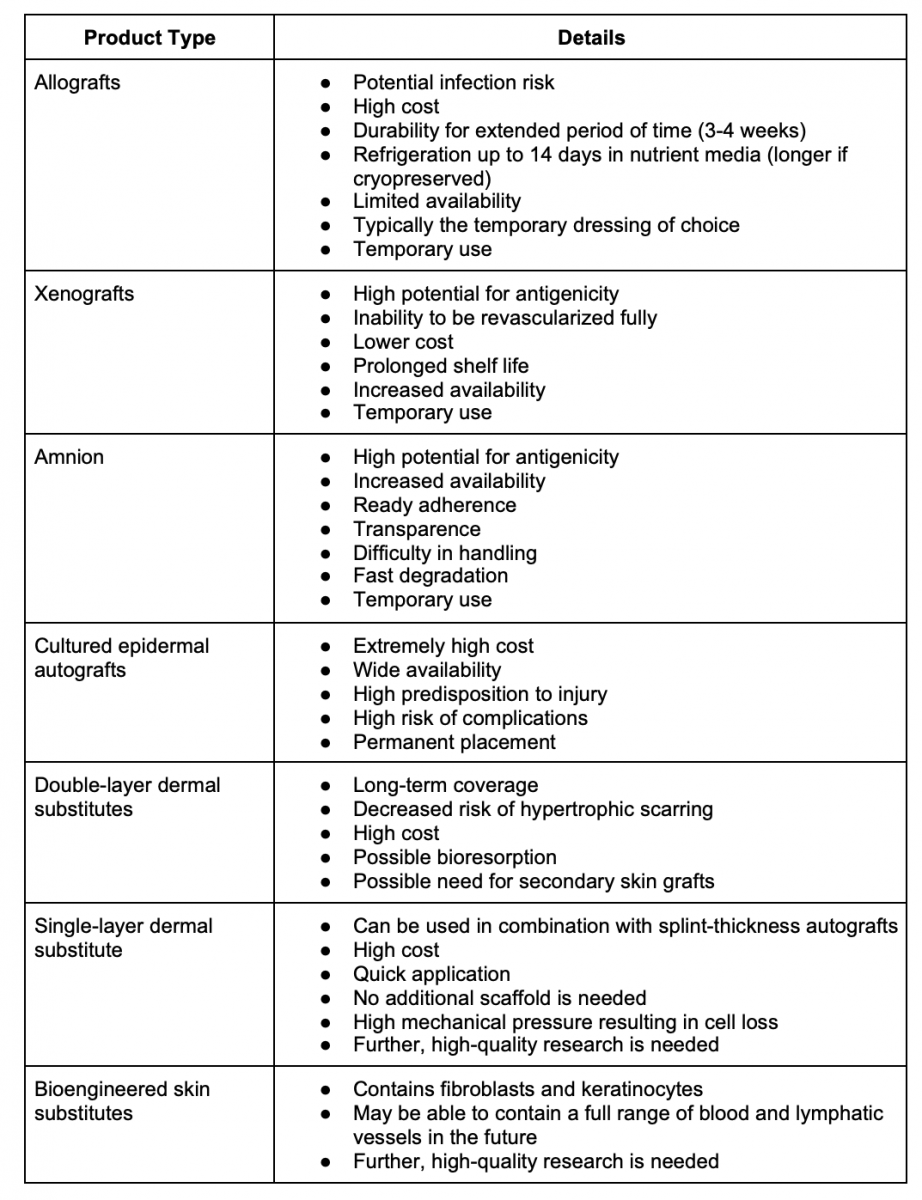
CTPs provide a variety of benefits for wound healing, including protection from infection and injury. Perhaps most importantly, however, CTPs act as a scaffold within the wound to provide transportation for cells and biomaterials that carry out the natural wound healing process.5 When a wound becomes too deep or too wide, the normal transport system is disrupted. Cells cannot migrate to areas of the wound that are no longer connected to the rest of the skin; therefore, a scaffold is needed. Currently, no CTP has emerged as a gold standard treatment for severe wounds and burns. Therefore, when choosing an appropriate product, practical aspects must be considered, including cost, storage, availability, and ease of application.6 Table 1 shows a comparison of CTPs.
Table 1. Comparison of Cellular and/or Tissue Based Products6
Conclusion
CTPs offer an important solution for wounds that are deep, wide, or otherwise difficult to heal. These products provide scaffolding that connects healthy tissue across the damaged area to promote healing and wound closure. CTPs should be considered by clinicians when wounds do not respond to other standard treatments or for treating burn wounds.
References
- Liu Y, Panayi AC, Bayer LR, Orgill DP. Current available cellular and tissue-based products for treatment of skin defects. Adv Skin Wound Care. 2019;32(1):19-25. doi:10.1097/01.ASW.0000547412.54135.b7
- Langer A, Rogowski W. Systematic review of economic evaluations of human cell-derived wound care products for the treatment of venous leg and diabetic foot ulcers. BMC Health Serv Res. 2009;9:115. doi:10.1186/1472-6963-9-115
- ASTM F3163-16: standard guide for classification of cellular and/or tissue-based products (CTPs) for skin wounds. West Conshohocken, PA: American Standards for Testing and Materials International; 2016. Accessed January 31, 2022. http://www.astm.org/cgi-bin/resolver.cgi?F3163-16
- Oberweis CV, Marchal JA, López-Ruiz E, Gálvez-Martín P. A worldwide overview of regulatory frameworks for tissue-based products. Tissue Eng Part B Rev. 2020;26(2):181-196.
- Dickinson LE, Gerecht S. Engineered biopolymeric scaffolds for chronic wound healing. Front Physiol. 2016;7:341. doi:10.3389/fphys.2016.00341
- Haddad AG, Giatsidis G, Orgill DP, Halvorson EG. Skin substitutes and bioscaffolds: temporary and permanent coverage. Clin Plast Surg. 2017;44:627-634.
The views and opinions expressed in this content are solely those of the contributor, and do not represent the views of WoundSource, HMP Global, its affiliates, or subsidiary companies.
More from this Author
Have a product to submit?
Be included in the most comprehensive wound care products directory
and online database.
Learn More










