The Impact of Hands on Education, Quality Incontinence Briefs and Wipes in Sub-Acute/LTC Facilities Study Overview
September 26, 2019
By: M. Sanantonio RN, C. Gunner RN, S. Trousaant LVN, M. Moore LVN
It is important for skilled nursing facilities to utilize quality incontinent products for their residents that are facing the daily challenges of both bladder and bowel incontinence. However, products alone do not make for a successful continence management program. It is imperative that the caregivers receive education in multiple areas that ultimately have an effect on the resident’s quality of life, quality outcomes and overall facility cost efficiencies. This study demonstrates the combination of quality incontinent products and education and its overall effectiveness in skilled nursing facilities.
I. Background
Incontinence is defined as the lack of voluntary control over urination and defecation. Urinary Incontinence (UI) can happen at any age affecting over 25 million Americans. “According to the National Association for Continence, one in five individuals over the age of 40 suffer from overactive bladder, urgency or frequency symptoms.” (Elderly Urinary Incontinence, 2015) Of those who are incontinent, one in four are women ages 30-59, and 50% or more are elderly living at home or in long term care (LTC) facilities. Studies have shown that the most frequently affected population is older adults.
Incontinence can be caused by diseases, infections, diet, medications, pregnancy, menopause, childbirth, and age. Aging causes body changes which can increase the likelihood of incontinence. According to Dr. Lisa Rosenberg, “Incontinence is a common part of aging, but is never normal.” (Elderly Urinary Incontinence, 2015) In a recent study it was estimated that approximately 25.6% of men and 48.4% of women residing in long term care settings were affected by incontinence that resulted in significant quality of life and dignity concerns.
While age is a large contributing factor in long term care settings, infections are as well. According to the CDC, the urinary tract is the most common site of healthcare associated infections. A urinary tract infection (UTI) is a very common infection in the bladder or urinary tract, and accounts for 20-30% of infections in long term care facilities. UTIs affect more women than men (about 40-50% of women will experience a UTI in their lifetime). While UTIs can be caused by many different types of bacteria, E.coli is the most common, causing about 85% of UTIs. E.coli bacteria lives in the entire bowel, including the rectum, and can lead to the development of a UTI when pushed into the urinary tract. This occurs when inappropriate wiping of the perineum occurs after a bowel movement. Cleansing of the perineum should occur by wiping from front to back, however staff and patients as well will wipe from back to front pulling the feces and the bacteria into the urinary tract.
There are several types of incontinence, which include stress, urge, overflow, functional, and mixed. Stress, one of the most common, occurs when urine leaks from the bladder when activity places pressure on the bladder, or when coughing or sneezing. This type occurs because of weakened pelvic floor muscles. Urge incontinence, also referred to as overactive bladder, can be caused by damage to bladder nerves or other muscles. Overflow incontinence occurs when the inability to empty the bladder results in dribbling of urine. This type of incontinence can be caused by nerve damage, weak bladder muscles, and even constipation. Functional incontinence, common in the elderly, can be caused by physical or mental limitations that can include dementia, or decreased mobility affecting the ability to get to the bathroom in time. Finally, mixed incontinence is a result of two or more different types of incontinence. For instance, many women will experience both urge and stress incontinence.
While there are various types of incontinence, many types have similar causes. Determining the type of incontinence requires an accurate assessment and the collaborative efforts of the entire interdisciplinary team. Treating and managing UI is a team effort and not the lone responsibility of nursing. Therefore, it is crucial for nursing and therapy to join together in promoting an effective continence program for facility residents. Statistics indicate a need for structured programs to decrease episodes of treatable incontinence, and recent outcome studies demonstrate a real-life improvement for those residents who receive therapy with interdisciplinary care team (IDT) involvement and programs.
There is increasing evidence of the need for effective management of the symptoms and conditions that contribute to incontinence. Yet in spite of this increasing evidence, it is common that many older patients do not receive an accurate assessment or appropriate treatment. While there are numerous advances that have been made in the treatment of incontinence, the unfortunate truth is that these advances are not routinely applied in practice, resulting in inaccurate assessments and untreated UI.
The lack of an accurate assessment and prolonged untreated UI can result in:
- Loss of self-esteem/dignity
- Altered or negative body image, decreased sense of well-being
- Embarrassment, anger, frustration, fear (of how others perceive them)
- Restriction of social activities and isolation
- Depression and guilt
- Loss of skin integrity, pressure injury risk
- Increased risk of falls
- Increased UTIs, infections
Of the many complications that can occur related to incontinence, one of the most concerning is the effect that incontinence has on skin integrity. Moisture Associated Skin Damage (MASD) and Incontinence Associated Dermatitis (IAD) result in problems for the patient as well as major challenges for healthcare staff, especially nursing. (Skin Irritation Related to Incontinence, n.d.) Managing skin damage creates numerous concerns for healthcare providers, as caring for these patients requires more time and can be more costly. Continued incontinence and lack of treatment and management can eventually lead to even more costly complications, including pressure injury development. Simply put, IAD is costly, painful, often misdiagnosed, undertreated, problematic, and a precursor to Pressure Injury (PI).
Because IAD is often confused with Pressure Injury, it is important that staff understand what IAD is and what causes it. IAD is the skin damage that is associated with exposure to body fluids, specifically urine and feces. IAD is also often referred to as intertrigo, perineal dermatitis, or diaper rash, and is listed with other skin conditions referred to as moisture-associated skin damage or MASD. IAD can be mild, moderate or severe.
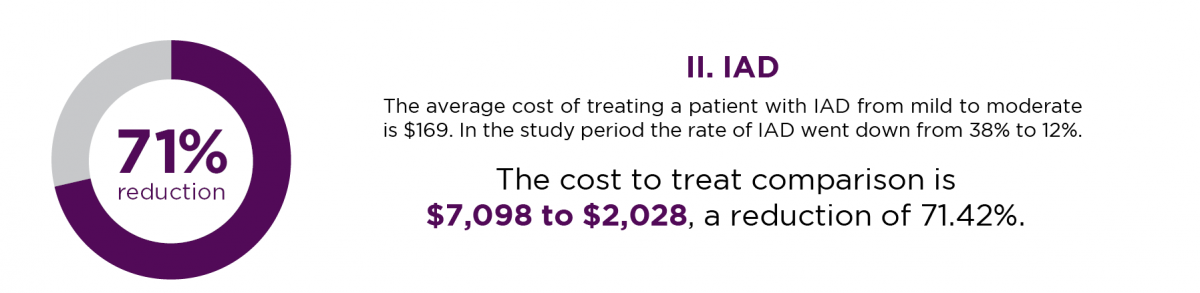
Factors including prolonged urine or feces exposure, altered skin pH, and use of incontinence devices all contribute to IAD. IAD is not caused by moisture alone. IAD occurs when the protective barrier created by your skin is damaged. Symptoms of IAD include: redness, patches of inflammation, warm and firm skin, lesions, pain, burning, and itching. The pattern of IAD is diffuse and the depth of skin damage is partial thickness. IAD is a common problem, creating legitimate concerns among practitioners based on recent studies. Failure to treat IAD as partial thickness skin damage — that in turn becomes full thickness — then becomes classified as a PI. A recent study showed that in an acute care setting 19.7% of incontinent patients had some level of IAD compared to the upwards of 50% of patients in a long term care facility. The economic impact of incontinence-associated dermatitis is significant. The inflation-adjusted annual estimated cost for skin conditions related to incontinence in the United States eclipses $215 million, and on average, in a long-term care setting, the cost to treat one mild to moderate IAD episode in one resident can be up to $169.
II. Initial Study Overview
An incontinence study was conducted in three facilities that have both SNF and Sub-Acute units in house. These facilities were labeled Facility “A,” Facility “B,” and Facility “C.” A blind draw was used to label facilities. The study team had no previous involvement or business with the facilities in the study. Products were unknown to the staff and sent in generic packaging. After study finalized, the products were disclosed as First Quality Products: Prevail Perfit 360 Adult Disposable Brief™, Prevail Ultimate Bladder Control Pads™, Prevail Premium Quilted Adult Washcloths™ and Prevail Bariatric “A” Briefs™.
- Facility “A:” Patients were assessed — however, there was no change in the product or the staff training and education. Facility “A” is our control facility.
- Facility “B:” Patients were assessed and there was the use of cloth backed disposable brief products, gender specific underwear, and incontinence wipes. The facility staff received no training or education.
- Facility “C:” Patients were assessed, cloth backed disposable briefs were used, wipes were provided, and floor and clinical staff received education upon initiation of the trial, with continued training throughout the duration of the trial period.
All incontinent patients in all buildings were initially assessed for sizing, skin conditions and type of incontinence, which resulted in a total of four hundred and twenty-four patients (424) with a median age of 68 years. Of the 424 patients, one hundred sixteen (116) were women and three hundred eight (308) were male. One hundred fifteen (115) or 27% of patients had some degree of IAD on initiation of study. Thirty-five (35) or 12% of the incontinent patients had a PI, and only PIs located in the brief area were counted. This study will look at the incidence of IAD/PI, and also assess the findings in each studied facility where there was a focus on education and training, as well as the use of new incontinence products, laundry costs and UTI rates.
Patient Breakdown
1. Patient Mix
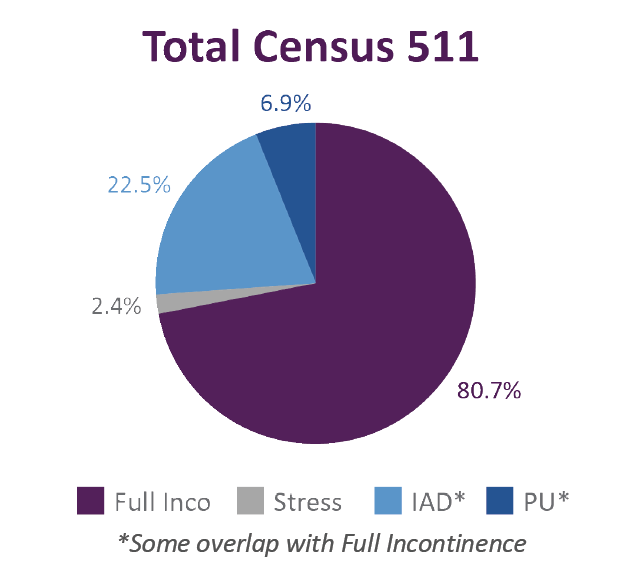
A total of 511 patients, with 412 patients having full incontinence, 12 patients having stress/urge incontinence, 115 patients with IAD, and 35 having a pressure ulcer in the incontinent brief area.
A. Pre-study facilities used a plastic backed brief for all buildings. The sizes were medium (M), large (L) and extra-large (XL). The vendor did not offer a bariatric brief. Larger patients were therefore placed in briefs that were too small, creating leakage, increased moisture on the skin, increased episodes of wet linens, and additional pressure if stretched to fit.
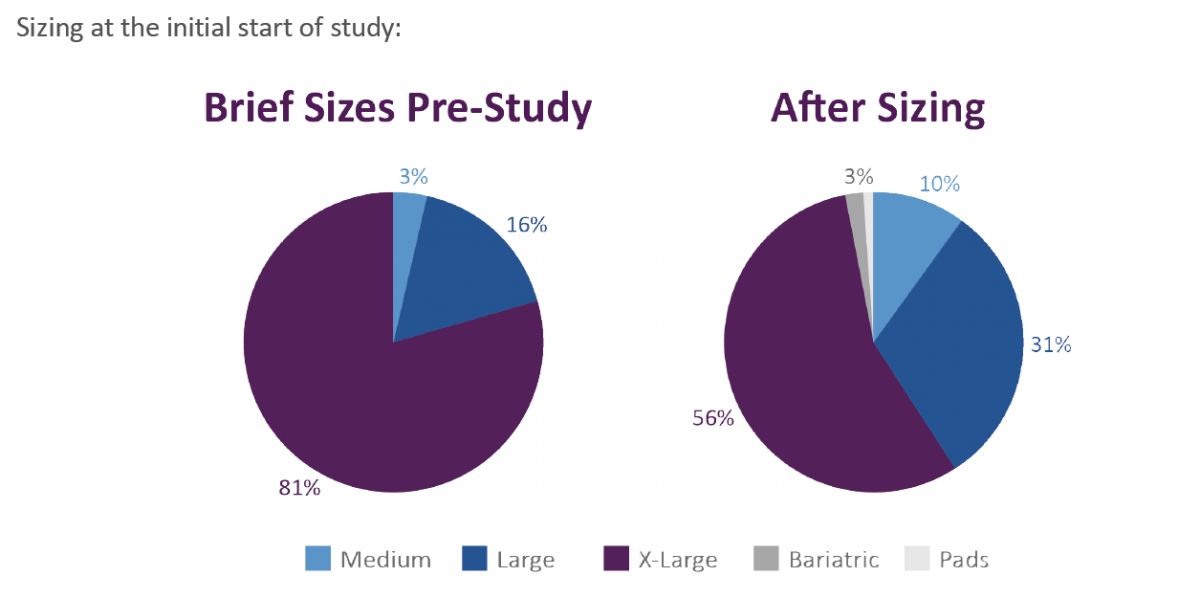
B. In each facility, there were a small percentage of patients, 2-4% that would have benefitted from the use of protective underwear; however, these were not offered in the facilities, so instead these patients were placed in full briefs. During the study, these patients were put in the protective underwear based on individual needs, allowing the patients to self-toilet.
C. The facilities utilized washcloths and antibacterial soap for providing incontinence care.
D. Laundry services were offered in the facility for personal clothing only, while facility linen was sent out to be laundered.
III. Study Roll Out
Prior to incorporation of the incontinence products, the following was completed by the study team. Skin assessed for conditions such as dryness, excoriation, denuded tissue and turgor. Briefs assessments were completed on all incontinent patients including the current size of brief vs. the size needed for the height and weight of the patient. Products were sized based on the “bigger is better” mentality. The staff felt that the larger briefs would hold more resulting in less changing. This thought process was a key topic in our educational topics.
- Facility “A:” 76% were in the wrong size brief.
- Facility “B:” 63% were in the wrong size brief.
- Facility “C:” 54% were in the wrong size brief.
The study staff completed a quality review at Facility “C,” and the facility also had full support, continued study staff, products, training, etc. throughout the study timeline. Overall the facilities quality review did not identify system failures with incontinence or skin care, as proper skin protocol was used, with appropriate wound treatments for types of wounds per guidelines.
Brief Training:
- The facilities were using briefs that weren’t properly fitting on many patients. The negative outcome of this was discussed with staff. Staff were further trained on the correct sizing, the action of polymer, the added work they are making for themselves and the financial impact.
- Staff were trained on proper turning and positioning, and cleansing properly using moistened wipes. Skin health and preventative measures were incorporated into training.
- Training also occurred regarding the proper use of incontinence cloth under pads, and how they shouldn’t be used as the primary turning and repositioning device.
Continued Education Areas:
- Patients were placed in chairs for extended periods of time. Study provided education regarding surface and repositioning, and the importance of changing the position of patients and placing them on different surfaces at different degrees.
- Staff used larger than necessary briefs during trial period, with the misconception that the bigger the brief the more urine it would hold. Staff were trained on properly sizing patients so that briefs were used appropriately.
- With many incontinence patients using the briefs, the staff were using the briefs to assist with turning. They used the briefs to pull patients up in bed and would use the briefs to upright patients. This behavior was modified during the study.
- The facility used washcloths instead of wipes for peri care. For those facilities that were given the wipes to use, education was provided to ensure proper use of the wipes.
IV. Post Study Findings
The study concluded after five weeks, the first week of data was discarded to ensure that no previous product info would alter data. Final skin assessments were completed, and the trial products were removed from building.
Procedural issues during trial period that changed with training, the bullet points shown below show that when front line staff is trained on key areas, they impact what was initially a negative response into a positive and stronger incontinence care program. This data was collected by both verbal and written questionnaire to the field front line staff.
- Trial brief absorbent quality: 36% at two weeks vs 14% at five weeks
- Side tearing briefs: 78% at two weeks vs 47% at five weeks
- Tabs tearing or not adhering: 57% at two weeks vs 20% at five weeks
- Brief leakage: 42% at two weeks vs 8% at five weeks
V. STUDY DATA VS. 30 DAY POST STUDY
After the product was pulled from the buildings, the previous brief was returned. The team made visits back to all buildings to conduct skin assessments, and follow up with staff for feedback.
A. Incontinent-Associated Dermatitis
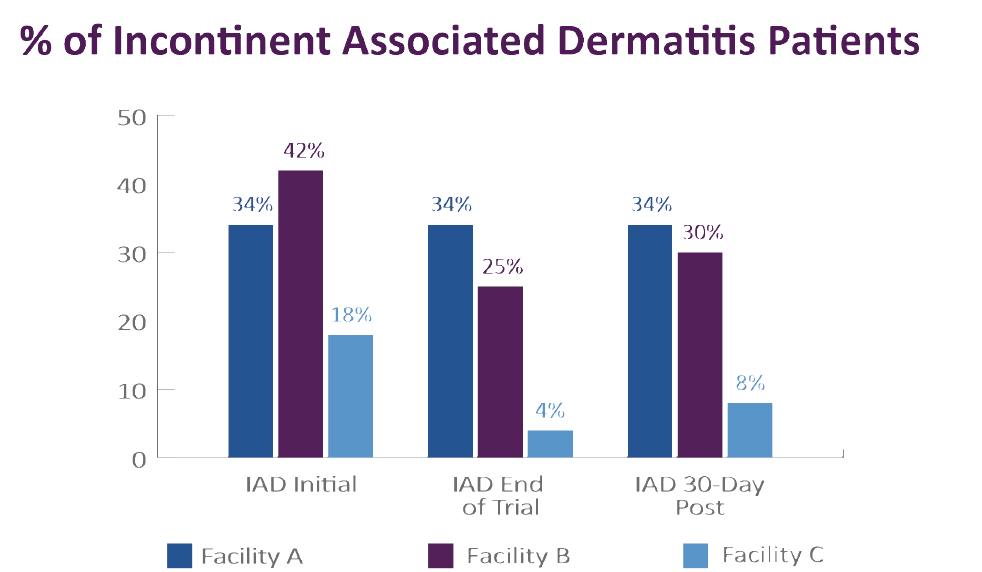
Incontinent-associated dermatitis was a factor in the buildings prior to the study with 115 total patients with mild to moderate Incontinent-associated dermatitis. The two facilities that had product in use showed a decrease of those affected from 38% at week 2 to 12% at week 5 during trial window but after the facilities went back to using soap and washcloths instead of wipes skin conditions increased to 17% in the non-trained facility (“B”) and to 4% in the trained facility (“C”).
The graph below shows the decrease of episodes when trial briefs, wipes and training protocol were used. After study ended and previous product, washcloths and soap were reintroduced, the skin conditions showed an upward trend of IAD.
B. UTI Rates
It is very common to have an increased in UTI when your population is incontinent and ensuring proper peri-care is needed. During the study, clinicians monitored the UTI rates.
- Facility “A” (control)
- September 6.0% (5/91)
- October 4.5% (4/91) — during trial period
- November 6.0% (5/91)
- December 6.0% (5/93)
- Facility “B” (product without training)
- September 5.0 (12/250)
- October 4.4% (11/249) — during trial period
- November 3.5% (9/255)
- December 2.8% (7/249)
- Facility “C”
- September 0.45% (1/224)
- October 0.45% (1/224) — during trial period
- November 2.2% (5/228)
- December 1.8% (4/228)
C. Financial Impact
Product costs for Facility “B’ and ‘C’ were compared after the trial was completed. Data compared the cost of products that would be a direct cross reference only. For example, if the facility did not pay for incontinent wipes before trial we did not include it in the financial comparison. The incontinent product cost of both facilities averaged $10,697 per month compared to the trial products of $8,266 which is a savings of $2,430 per month or annualized is a savings of $29,160 or reduction of 28%.
Personal clothing was laundered in the facility, but all linens and cloth incontinence pads were sent out to be laundered by a service (“Month 2” is study window). Previous products absorbency and incorrect sizing were challenges that impacted the need for complete bed linen changing. At the onset of the study, up to every other brief change required full linen changes during incontinent episodes. During study, education on sizing and proper fitting were initiated and full bed changes decreased to 10% by end of study. The costs savings were $5,920 for linen fees. Noted costs returned to pre-study costs after trial products and educators were removed.
- Facility “A”
- Month 1 - $24,449
- Month 2 - $21,975-study period
- Month 3 – $23,653
- Facility “B”
- Month 1 - $34,325
- Month 2 - $32,796-study period
- Month 3 - $33,740
- Facility “C”
- Month 1 - $21,901
- Month 2 - $15,981-study period
- Month 3 - $20,968
D. Sizing Usage Of Patients
Sizing usage in the non-trained building: Facility “B” – The sizing usage went back to 78%-XL and 22%-L. They stopped using M, and no pads or bariatric briefs were used.
Sizing in the trained building: Facility “C” – The sizing stabilized at 44%-XL, 40%-L, 12%-M, and the remaining 4% included bariatric briefs and pads.
Stress/Urge patients: There was a total of 3% of the incontinence patients in all facilities with stress or urge incontinence that initially were in full brief, regardless of amount of incontinence. After training for both patient and staff, 66% of those patients were taken out of briefs and bladder control pads were utilized. Patient satisfaction: All facilities had only had three sizes to choose from previous to the trial. The trial provided bariatric sized briefs. In Facilities “B” and “C,” 100% of the bariatric patients found proper sizing for better fit and appropriate absorption for incontinence. Clinical team obtained feedback directly from the patients when allowed, both verbal and written questionnaires were utilized.
Staff Comments
Clinical Team obtained feedback from staff via a written questionnaire at week 2 and at week 5:
- Sizing swing. The sizes were not properly fitting, even when sized by study group. Staffed was educated on unisex briefs and sizing needs, we also looked at the patient and made brief decisions on the patient’s body structure.
- Sides are weak. Sides did not hold up as well. Training occurred regarding not using the brief as a form of repositioning tool.
- Men’s briefs should have more padding in the front. The briefs did not have the required absorbency in the areas needed based on gender. Training on proper placement of genitals to ensure that voiding would be in the area of polymer.
- The briefs were too bulky between the legs and not comfortable. Training occurred on proper application of brief and positioning it correctly between legs.
- Body shape changes by gender e.g. a five-foot tall 180 pound woman is shaped differently than a man of the same height and weight, which affects correct sizing. Discussion on knowing your patient and use the right product size for them that address the amount of void, body shape and patient specific needs.
Consultant Comments
The facilities have challenges that are not unique to the facilities, but rather industry-related issues.
- Staff-to-patient ratio on all shifts. All three facilities are located in a major metropolitan community that is inundated with medical and nursing facilities that pull from the pool of staff. Ratios of the pt. per nurse is at the state ratio, and some days over the state ratio.
- Turnover. With heavy competition of facilities vying for the pool of staff, turnover is at an all-time high, ranging from 55% to 75% with some areas as high as 100%. The facilities had to implement creative marketing and recruiting programs to interest staff in employment. One such creative program was implemented in one of the facilities during the trial period. Facility “C” offered a promotion that included the giveaway of a car to any floor staff that stayed for six (6) months, and maintained an exceptional employment record during that time, which included no call offs and no tardiness. Unfortunately, the car wasn’t enough of an incentive to keep employees, as there were no employees who achieved the employment requirements to receive the car during the period of the study.
- Training. With the high rate of turnover, training was a difficult prospect. Staff changed so often in the facilities, training would literally have had to take place on a daily basis, across all shifts, in order to keep enough adequately-trained staff on the floors to care for the residents. The constant attrition makes onetime training insignificant.
VIII. Conclusion
The product cost savings, decrease in IAD, laundry costs and continuity in care shows that the impact on both medical and financial costs. Incorporating a high quality breathable disposable brief, incontinence wipes, education, and training are positive for facilities; however, the challenges that facilities face with staffing, patient ratios, and a need for continued training with reinforcement may create challenges for expectations of manufacturers and customers alike.
Additional Statistics
I. Overview
A 30-day incorporation of incontinent products including briefs, pads, and incontinent wipes with skin and wound training, turning and repositioning and sizing. The areas of focus looked at costs for correct sizing, laundry costs and skin condition costs. The economic impact of incontinence-associated dermatitis is significant. The inflation-adjusted annual estimated cost for skin conditions related to incontinence in the United States eclipses $215 million, and on average, in a long-term care setting, the cost to treat one mild to moderate IAD episode in one resident can be up to $169.


Bibliography
Beeckman D, Verhaeghe S, De oor T, Schoonhoven L, Vanderwee K. A 3-in-1 perineal care washcloth impregnated with dimethicone 3% versus water and pH neutral soap to prevent and treat incontinence-associated dermatitis: a randomized, controlled clinical trial. J Wound Ostomy Continence Nurs.2011;38(6):627-634.
Beeckman D et al. Proceedings of the Global IAD Expert Panel. Incontinence-associated dermatitis: moving prevention forward. Wounds International 2015. Available to download from www.woundsinternational.com.
Definition of Incontinence (n.d.) Merriam-Webster Dictionary. Retrieved from https://www.merriam-webster.com/dictionary/incontinence.
Elderly Incontinence: Causes and Care. (2015, April 15). https://www.aplaceformom.com/senior-careresources/articles/elderly-urin….
Long, M., Reed, L., Dunning, K., Ying, J. (2012, May-June). Incontinence-associated dermatitis in a longterm acute care facility. Journal of Wound Ostomy Continence Nursing. Retrieved from: https://www.ncbi.nlm.nih.gov/pubmed/22552105.
Minassian V, Devore E, Hagan K, et al. Severity of urinary incontinence and effect on quality of life in women, by incontinence type. Obstet Gynecol. 2013; 121(5): 1083-90.
National Pressure Ulcer Advisory Panel, European Pressure Ulcer Advisory Panel and Pan Paci c Pressure Injury Alliance. Prevention and Treatment of Pressure Ulcers: Quick Reference Guide. Emily Haesler (Ed.). Cambridge Media: Osborne Park, Western Australia; 2014.
Skin Irritation Related to Incontinence: How to Prevent it and Manage it in Your Patients: White Paper. (n.d.). Convatec. Retrieved from: https://kestrelhealth.lpages.co/skin-irritation-related-to-incontinence….
Urinary Tract Infection (UTI) Event for Long-term Care Facilities. Centers for Disease Control. Retrieved from https://www.cdc.gov/nhsn/pdfs/ltc/ltcf-uti-protocol_final_8-24-2012.pdf.
The cost of turnover in nursing homes. McKnight’s 2010.
About First Quality
First Quality Products, Inc., the makers of Prevail Adult Incontinence products, was founded in 1988, and is a leading manufacturer in the adult incontinence health care industry. First Quality Products is a member of the First Quality Group of companies, a diversified family of companies manufacturing adult incontinence, baby, feminine hygiene and paper products. For information about First Quality, their products and clinical support please visit: www.prevail.com.
The views and opinions expressed in this blog are solely those of the author, and do not represent the views of WoundSource, Kestrel Health Information, Inc., its affiliates, or subsidiary companies.
The views and opinions expressed in this content are solely those of the contributor, and do not represent the views of WoundSource, HMP Global, its affiliates, or subsidiary companies.






