Surgical Site Infections: Current Recommendations and Guidelines
December 8, 2017
Great news! Data suggest that surgical site infection (SSI) incidence could be halved with implementation of evidence-based interventions. So, why are interventions not ubiquitously utilized across health care institutions and SSIs not nearly eradicated? One complicating factor is that there is not yet one organization or document with a complete, finite consensus of measures to prevent or decrease the incidence of SSIs. Multiple organizations and regulatory bodies (American College of Surgeons [ACS], American Society of Colon and Rectal Surgeons [ASCRS], Association of periOperative Registered Nurses [AORN], Centers for Disease Control and Prevention [CDC], The Joint Commission [TJC] – formerly the Joint Commission on Accreditation of Healthcare Organizations [JCAHO], World Health Organization [WHO], etc.) have recommendations or guidelines for prevention, and most have several unifying components.
Most consensus statements, recommendations, and comprehensive guidelines address patient-specific risk factors, as well as interventions aimed at the prehospital, hospital, and post-discharge stages of care. Although a multitude of similarities exists, there are notable variances. These can be attributed to differences in data interpretation, regard for target audience, and priorities for a given organization. There exists a need for a national consensus with support from multiple organizations with input from all disciplines and specialties to determine absolute best practices for prevention of SSI.
Grouped Interventions to Control Surgical Site Infections: Bundles
Many health care and regulatory organizations have launched efforts aimed to affect the occurrence of SSIs. Some of these efforts have included various basic practices that most institutions already utilize, whereas others have recommended the adoption of special approaches for when basic practices have not been sufficient in controlling SSIs, commonly referred to as "bundles." Bundles are grouped interventions deployed at predetermined intervals throughout the stages of surgical care. Many hospitals utilize bundles validated through national organizations, as well as those adopted by corporate entities that govern multiple facilities in various geographic locations. One prominent theme with bundles relates to provider and patient compliance; multiple high-quality studies demonstrate mixed data concerning bundles, thus emphasizing that provider buy-in and patient compliance are requisite for success.
Surgical Site Infection Guidelines
I will review the two most recent and comprehensive guidelines: the WHO Global Guidelines for the Prevention of Surgical Site Infections (SSI) 2016; and the American College of Surgeons and Surgical Infection Society (SIS): Surgical Site Infection Guidelines, 2016 Update. The WHO guidelines focus on 29 topics relative to preoperative, intraoperative or perioperative, and postoperative patient care interventions. A systematic review is provided for each intervention, which is then graded based on the strength and quality of evidence available. The ACS guidelines contain 22 general recommendations, and they were developed by qualitative analysis and review of the available literature for gaps in current knowledge relative to previous guidelines. For comparison purposes, the Centers for Disease Control and Prevention (CDC) guidelines are also reviewed here. As stated earlier, the wording of guidelines may differ because of variances among objectives and methods.
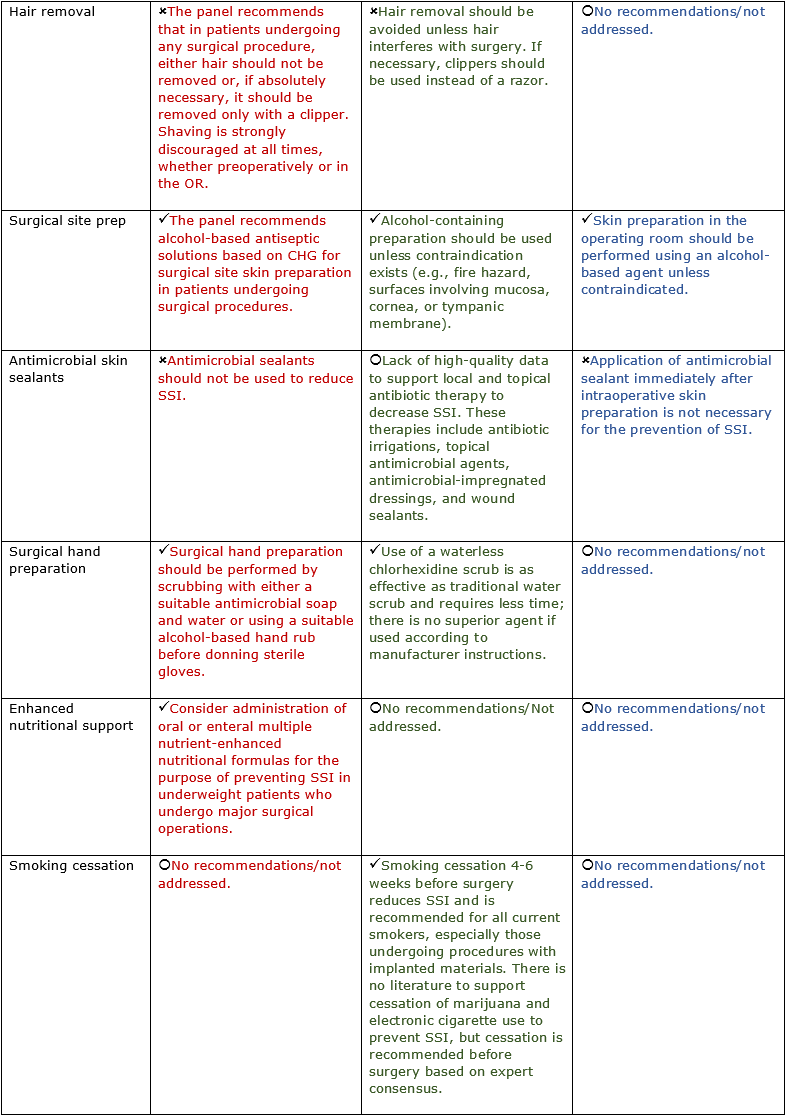
Preoperative/Prehospital
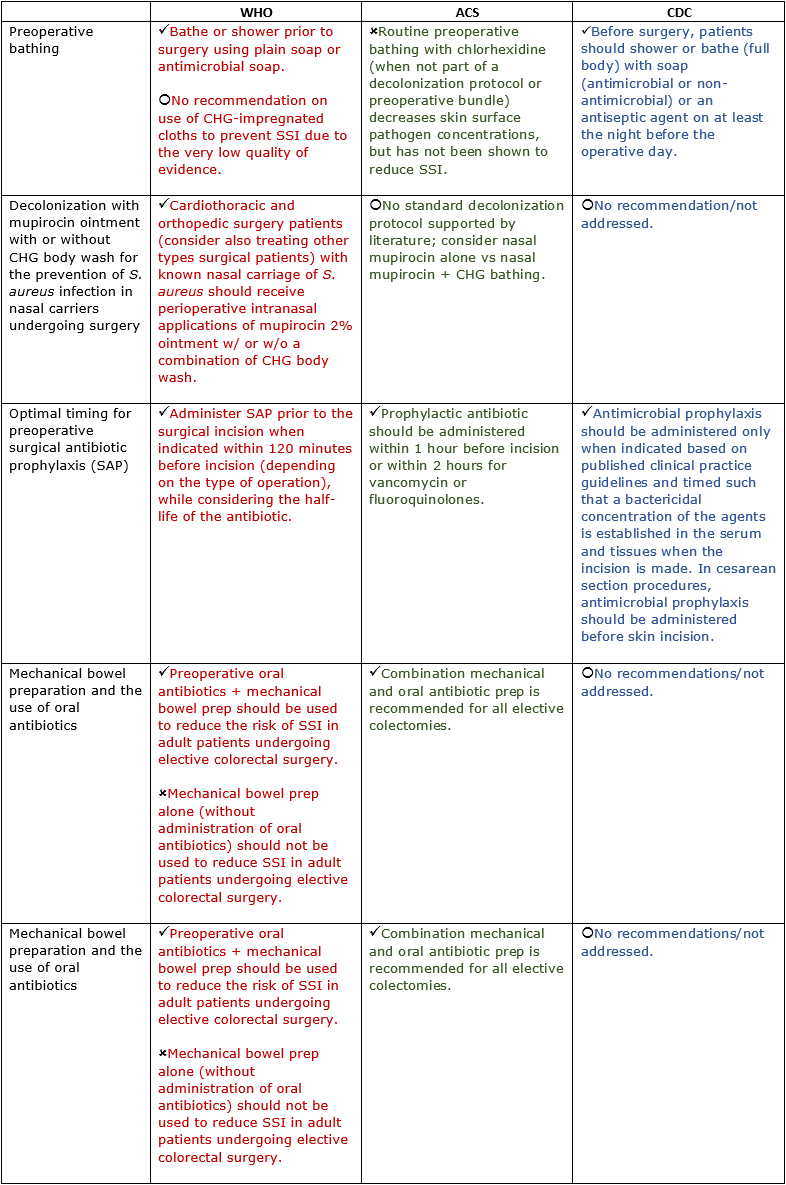
Intraoperative/Perioperative/Hospital
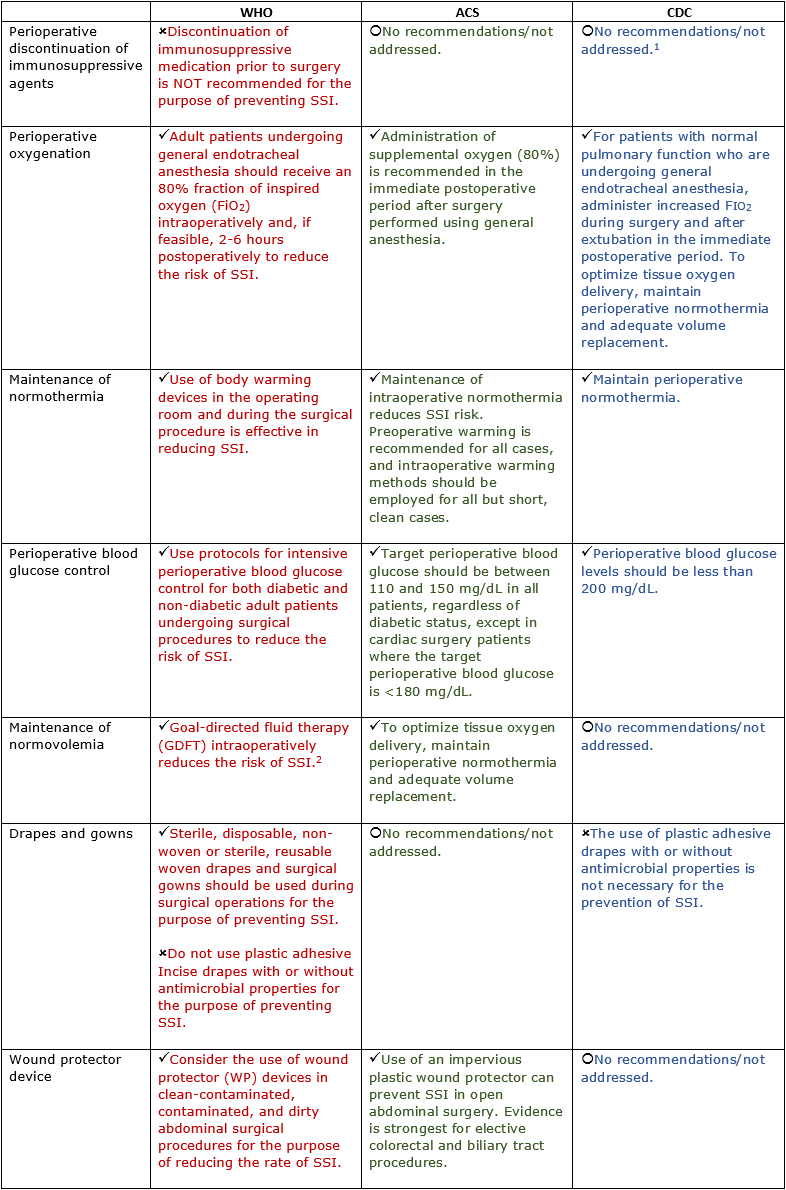
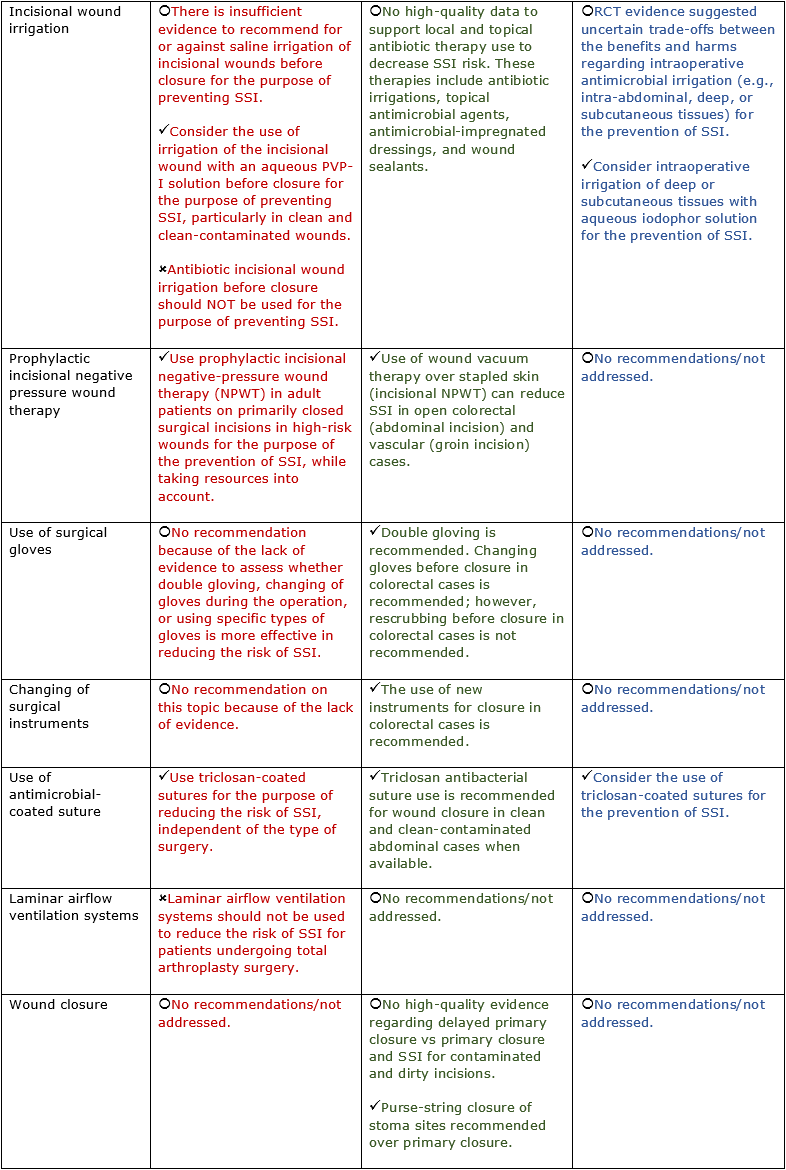 1In 2008, ACR recommended that biologic agents not be used for at least 1 week before and 1 week following surgery (based on the pharmacokinetic properties of a given agent). The 2012 ACR update does not address perioperative management of immunosuppressive therapy. ACS, American College of Surgeons; CDC, Centers for Disease Control and Prevention; CHG, chlorhexidine gluconate; SSI, surgical site infection; WHO, World Health Organization. (Centers for Disease Control and Prevention. Guideline for the Prevention of Surgical Site Infection 2017: Background, Methods, and Evidence Summaries.) 2GDFT refers to a hemodynamic treatment based on the titration of fluid and inotropic drugs according to cardiac output or similar parameters.
1In 2008, ACR recommended that biologic agents not be used for at least 1 week before and 1 week following surgery (based on the pharmacokinetic properties of a given agent). The 2012 ACR update does not address perioperative management of immunosuppressive therapy. ACS, American College of Surgeons; CDC, Centers for Disease Control and Prevention; CHG, chlorhexidine gluconate; SSI, surgical site infection; WHO, World Health Organization. (Centers for Disease Control and Prevention. Guideline for the Prevention of Surgical Site Infection 2017: Background, Methods, and Evidence Summaries.) 2GDFT refers to a hemodynamic treatment based on the titration of fluid and inotropic drugs according to cardiac output or similar parameters.
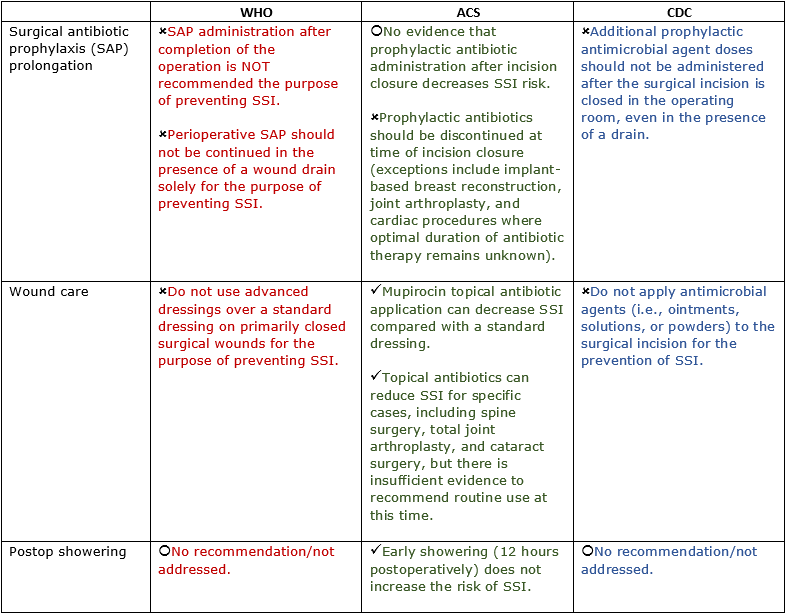
Postoperative/Posthospital
 CDC Takeaway: Compilation of the guideline revealed gaps, which fueled specific topics for future research. More research and technological advancement in the field of surgery will beget further development and honing of guidelines.
CDC Takeaway: Compilation of the guideline revealed gaps, which fueled specific topics for future research. More research and technological advancement in the field of surgery will beget further development and honing of guidelines.
ACS Takeaway: Guidelines are a framework for building tailored evidence-based care. Stakeholder buy-in is paramount to successful implementation of interventions and subsequent improvement in SSI rate. Education and provider involvement are key.
WHO Takeaway: Presenting data specifically for the target audience will yield best results. National norms do not always translate well to local needs. Adapting guidelines to the needs of individual organizations is necessary for best implementation and subsequent reduction in harm with regard to SSI and sequelae. What does this information mean for us as wound specialists? Because there is not currently a "one size fits all" solution for SSI prevention, we must give as much effort in the expert treatment of wounds as we do in enacting preventive measures. The current consensus among all organizations focuses on prevention. There is a paucity of research on wound care in the posthospital setting. Additionally, no formal wound care protocol exists that has proven to decrease SSI risk.
Recent Literature of Interest
A recent article published in Ostomy Wound Management addresses the possibility of grouping acute and chronic wound infection surveillance utilizing a multidisciplinary, evidence-based wound infection prevention and treatment guideline: http://www.o-wm.com/article/development-and-content-validation-multidis… It is worth noting that the surveillance period (essentially the timeframe for identifying a postoperative wound complication as a true SSI) varies by procedure, from 30 to 90 days. In addition, a superficial SSI is followed only for a period of 30 days regardless of procedure type. For specific procedures that involve secondary incisions, the primary incision may require a longer surveillance period than the secondary incision. An example would be the sternal incision (primary site) for a coronary artery bypass graft and the saphenous vein harvest incision (secondary site).
References
Ban KA, Minei JP, Laronga C, et al. American College of Surgeons and Surgical Infection Society: surgical site infection guidelines, 2016 update. J Am Coll Surg. 2016;224(1):59–74. Available at: http://www.journalacs.org/article/S1072-7515(16)31563-0/pdf
Berríos-Torres SI. Surgical site infection (SSI) toolkit. Centers for Disease Control and Prevention. 2009. Available at: https://www.cdc.gov/HAI/pdfs/toolkits/SSI_toolkit021710SIBT_revised.pdf
Berríos-Torres SI, Umscheid CA, Bratzler DW, et al. Centers for Disease Control and Prevention guideline for the prevention of surgical site infection, 2017. JAMA Surg. 2017;152(8):784–91. Available at: https://www.americannursetoday.com/jamasurgery
Centers for Disease Control and Prevention. Surgical site infection (SSI) event. 2017. Available at: http://www.cdc.gov/nhsn/PDFs/pscmanual/9pscssicurrent.pdf
Umscheid CA, Mitchell MD, Doshi JA, Agarwal R, Williams K, Brennan PJ. Estimating the proportion of healthcare-associated infections that are reasonably preventable and the related mortality and costs. Infect Control Hosp Epidemiol. 2011;32(2):101–14.
World Health Organization. Global guidelines for the prevention of surgical site infection. Available at: http://apps.who.int/iris/bitstream/10665/250680/1/9789241549882-eng.pdf…
About the Author
Samantha Kuplicki is board certified in wound care by both the American Board of Wound Management as a Certified Wound Specialist (CWS) and by the Wound, Ostomy and Continence Certification Board as a Certified Wound Care Nurse (CWCN) and Certified Foot Care Nurse (CFCN). She serves on the American Board of Wound Management (ABWM) Examination Committee and also volunteers for the Association for the Advancement of Wound Care.
The views and opinions expressed in this content are solely those of the contributor, and do not represent the views of WoundSource, HMP Global, its affiliates, or subsidiary companies.