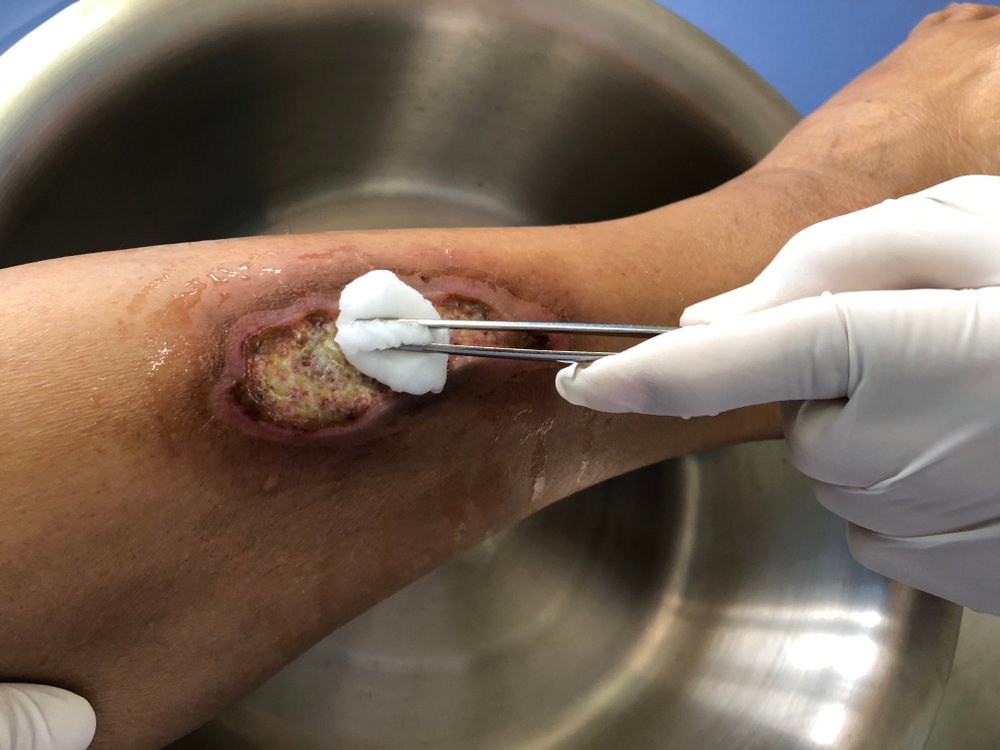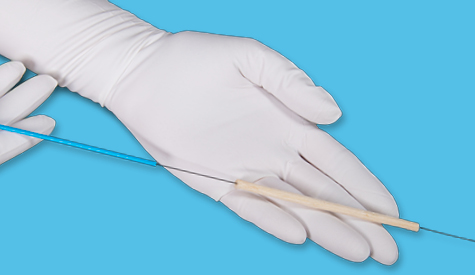
MiroTract™
MiroTract™ wound matrix consists of a compressed segment of dry decellularized porcine liver that is loaded onto a guidewire along with a plastic tamp tube and a stopper disc. For user convenience, the guidewire and tamp tube provide a delivery system for the wound matrix to be placed into a wound bed or tract, such as a crack, crevice, undermining, and tunneling space. If using the guidewire and tamp tube delivery system, after the guidewire position is established, the user slides the tamp tube down the guidewire to the distal end of the wound matrix and then pushes the matrix off the wire and into the wound bed. Once in place, the guidewire and tamp tube are removed and only the wound matrix remains within the wound bed. Alternatively, the wound matrix may be removed from the guidewire and delivered manually into the wound bed.
MiroTract™ contains a separately packaged optional tearaway introducer set. The introducer set is comprised of a dilator and an 8cm long tearaway sheath. If desired, the tearaway introducer set can aid in delivering the wound matrix to the desired location in the wound bed, however it is not required. Once the introducer set is positioned, the dilator is removed, and the wound matrix is delivered through the in-place sheath. The tearaway sheath is then removed along with the guidewire and tamp tube while the matrix remains in the wound bed.
Reprise Biomedical
Reprise Biomedical is focused on the development and commercialization of wound care matrices that offer advanced solutions for healing. Their products are derived from porcine liver utilizing a patented perfusion decellularization technology.Benefits
• MiroTract™ is designed for delivery to tunneling wounds or undermining wounds
• Porcine-derived collagen wound matrix compressed on a guidewire
• Expands and relaxes when hydrated for conformity within wound beds
• Porous 3D structure for full wall apposition
Indications
MiroTract™ wound matrix is intended for the management of wounds including partial and full thickness wounds, pressure ulcers, venous ulcers, chronic vascular ulcers, diabetic ulcers, tunneled, undermined wounds, trauma wounds (abrasions, lacerations, partial thickness burns, and skin tears), draining wounds, surgical wounds (donor sites/grafts, post-Mohs surgery, post-laser surgery, podiatric, wound dehiscence).
Contraindications
This device is derived from a porcine source and should not be used in patients with known sensitivity to porcine material.
This device is not indicated for use in third-degree burns.
Warnings and Precautions
MiroTract™ is supplied sterile for single-use only. Reuse of a single-use device creates a potential risk of patient or user infections and may compromise the device functionality, which may lead to illness or serious injury.
Do not resterilize, as the safety and performance has not been evaluated for this scenario. This is a single-use device. Reuse of this device creates a potential risk of patient infections.
Do not use a device past the expiration date as the safety and performance has not been evaluated for this scenario.
Do not use if the package or seal is opened, damaged, or compromised. A damaged package could result in a breach of sterility or device damage, which may lead to illness or serious injury.
Do not use the device on persons with a hypersensitivity or allergy to nickel or titanium as the nitinol wire component of the device contains these alloys and may create a potential allergic response.
After use, handle and dispose of all unused product and packaging in accordance with accepted medical practice and applicable local, state, and federal laws and regulations. This is a single-use device. Reuse of this device creates a potential risk of patient infections.
Adverse Effects/Reactions
• Allergic reaction
• Excessive redness, pain, swelling, or blistering
• Fever
• Infection including abscess
• Chronic Inflammation
• Non-healing wound
Potential adverse events only related to wire or introducer set:
• Device dislodgment
• Device fracture or separation
• Inflammation
• Necrosis
• Pain
• Scarring
• Tissue puncture or perforation
Storage Requirements
MiroTract™ is a sterile medical device that should be stored in a clean, dry location at room temperature, in its original package. Avoid prolonged exposure to elevated temperatures as it may compromise device functionality.
The product expiration date is indicated as year (4 digits), month (2 digits), and day (2 digits).
How Supplied/Sizing
HCPCS Code
Product features
Other features
Recommended Use
Acute Wounds
Chronic Wounds
Deep Wounds
Diabetic Foot
Necrotizing Fasciitis
Pressure Ulcers
Venous Ulcers
Mode of Use/Application
Prepare the wound using standard clinical practice methods ensuring that the wound is clear from debris and devitalized tissue. An initial debridement of the wound may be necessary to ensure the wound edges contain viable tissue. Follow standard clinical and hospital protocol.
If using the tearaway introducer set for deployment, follow steps
1. Measure the initial size of the wound, including depth, using standard clinical practice methods (e.g.; long cotton tipped applicators or surgical probe). Note the depth of the wound in relation to the overall length of the guidewire to assist in delivery of the device to the terminal end of a tunnel space or blinded wound bed.
2. Place the tearaway introducer set in the desired wound location.
3. Once the sheath is in the desired location, remove the dilator.
4. Locate the guidewire and wound matrix system, remove and discard the stopper disc from the distal end of the tamp tube.
5. Insert the guidewire with the wound matrix and tamp tube through the lumen of the sheath.
6. Ensure the stopper disc is removed from the end of the wire. Then, using aseptic technique, navigate the tip of the guidewire to the desired terminal end of wound.
7. Slide the tamp tube to the distal end of the wound matrix and gently push the matrix off the guidewire and into the wound. MiroTract™ should be placed in maximum possible contact with healthy, well-vascularized tissue as a scaffold to provide a protective environment for the wound.
8. If using the tearaway introducer set, grasp the butterfly hub and separate it. Gently peel the tearaway sheath down, exposing the tamp tube, guidewire, and placed wound matrix. Alternatively, the sheath can be removed without using the tearaway function. Carefully remove the remaining tearaway sheath, tamp tube, and guidewire, leaving the wound matrix in place.
9. Using sterile scissors or scalpel, carefully cut away any excess wound matrix outside of the wound space.
10. Using sterile saline or Lactated Ringer’s, flush the wound containing the wound matrix to allow it to hydrate, relax, and conform to the wound bed.
11. Use an appropriate non-adherent primary wound dressing over MiroTract to maintain matrix adherence and protect the wound area.
12. Apply an appropriate secondary dressing that will manage the wound exudate, keep MiroTract moist, and securely in place.
13. Discard the wire, tamp tube, stopper disc, sheath/dilator, and packaging in accordance with accepted medical practice and applicable local, state, and federal laws and regulations.
14. Per standard clinical protocol, the patient should be monitored by trained clinicians to full resolution of the wound using the minimum standard follow-up intervals of every seven to fourteen days with in-person visits for physical examination of the target wound.
15. As healing occurs, sections of MiroTract™ may not integrate. Carefully remove any remaining loose product as needed. Do not remove any remaining MiroTract™ that has integrated.
16. If the wound is free of infection and necrosis but not fully epithelialized, follow standard clinical protocol for additional application or therapy.
No posters match your selected filters. Remove some filtres, or reset them and start again.
Have a product to submit?
Be included in the most comprehensive wound care products directory
and online database.
Learn More